
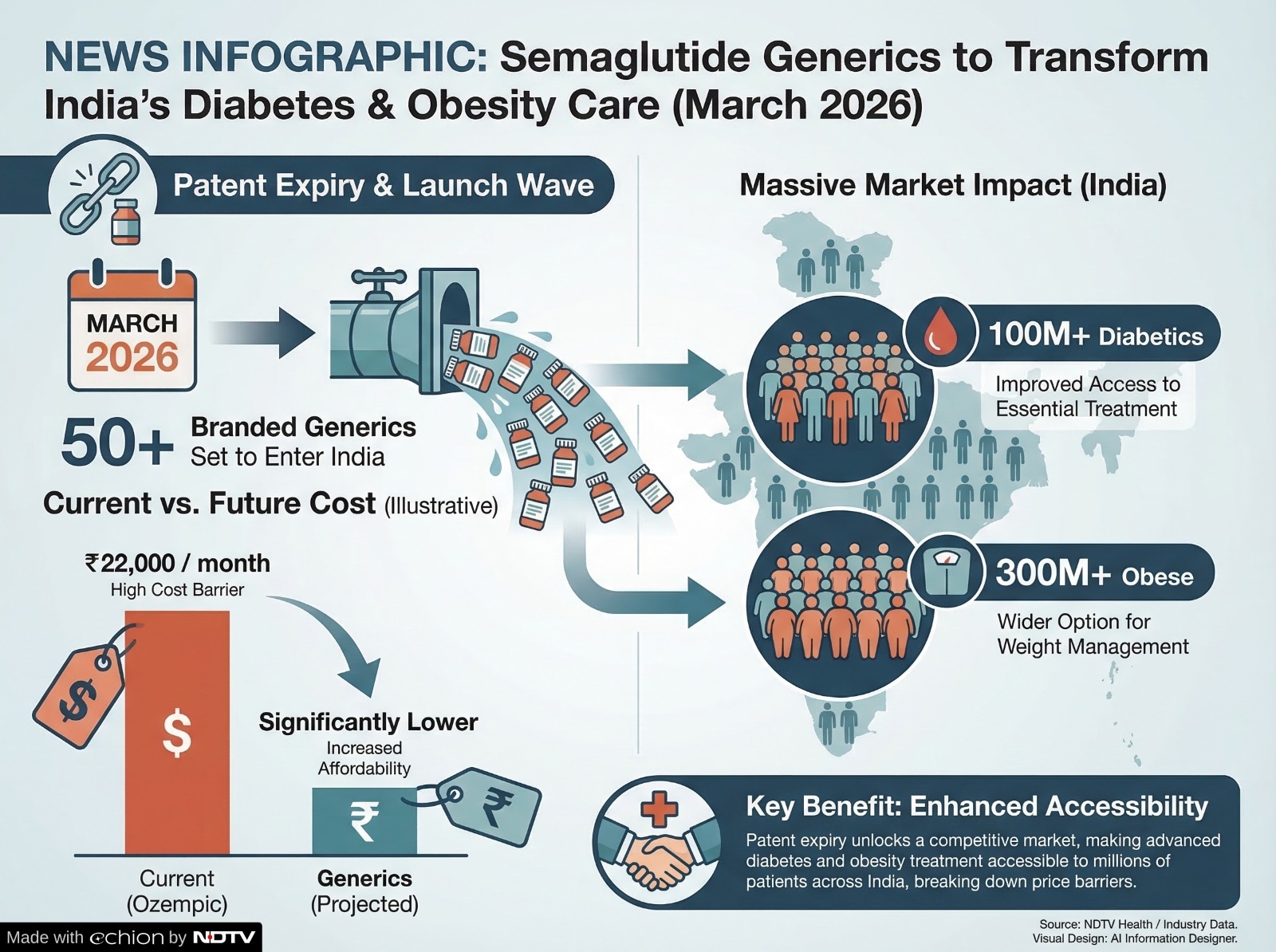
- Semaglutide patent in India expires on 20 March 2026, enabling generics entry
- Over 50 branded generic semaglutide versions expected to enter the Indian market
- Generics may cost around Rs 5,000 weekly, nearly 50% cheaper than branded drugs
The upcoming expiry of the semaglutide patent in India in mid-March 2026 is expected to transform the country's diabetes treatment landscape. Ahead of this, industry experts predict that the development will open the doors for more than 50 branded generic versions of the drug to enter the market, which may intensify the competition and potentially lower the overall treatment costs for patients. The main patent on semaglutide, the active ingredient in blockbuster drugs like Ozempic and Wegovy developed by Novo Nordisk, is set to expire in India on 20 March. Semaglutide belongs to a class of drugs known as GLP-1 receptor agonists and is widely used in the treatment of type 2 diabetes. In recent years, it has also gained global recognition for its role in weight management. Originally developed by Novo Nordisk, semaglutide is marketed globally under well-known brands used for both diabetes and obesity management.
What Makes Semaglutide So Important
Semaglutide functions by mimicking the glucagon-like peptide-1 (GLP-1) hormone, which plays an important role in regulating blood sugar levels. It stimulates secretion of an important hormone called insulin, suppressing the glucagon release. This slows down the gastric emptying and reduces appetite. These mechanisms help patients control blood glucose levels while also contributing to weight loss.
Also read: Weight-Loss Drugs Set To Get Cheaper In India As Semaglutide Patents Expire
Because of its dual benefits for both diabetes and obesity management, semaglutide has become one of the most sought-after therapies globally. However, the relatively high cost of branded versions has limited access for many patients, particularly in price-sensitive markets such as India.
Over 50 Branded Generics Expected
According to industry estimates, the patent expiry scheduled for mid-March 2026 will allow numerous domestic pharmaceutical companies to introduce their own branded generic versions of semaglutide. Experts believe that more than 50 brands could enter the Indian market soon after the patent protection ends.
As India's pharmaceutical sector is known for its strong generic manufacturing capabilities, many domestic drugmakers have already been preparing for the patent cliff by developing their own semaglutide formulations so that they can launch products as soon as regulatory pathways allow.
Major Pharma Companies Preparing for Launch
Several Indian pharmaceutical companies are expected to participate in the race to launch generic semaglutide injections. Companies such as Dr Reddy's Laboratories, Sun Pharmaceutical Industries, Cipla, Zydus Lifesciences, Lupin, Mankind pharma, Ajanta Pharma, Sun Pharmaceutical Industries, Lupin etc. are among those likely to introduce their own versions of the drug.
These generic alternatives could cost around Rs 5,000 per weekly dose, roughly 50% cheaper than the original therapy. The price drop is expected to significantly widen access to anti-obesity medication in a country where most patients pay for healthcare out of their own pocket. Some manufacturers are also working on user-friendly delivery systems such as pre-filled injection pens. These devices can make treatment easier for patients and may improve adherence to long-term therapy.
Also read: People Continue Taking Ozempic For Weight Loss Despite Side Effects, Reveals Study
Despite the anticipated entry of dozens of brands, experts suggest that only a few companies are likely to capture a major share of prescriptions. In most cases, firms with strong distribution networks, physician relationships, and brand recognition tend to dominate the market over time.
Patent Expiry Opens The Door To Cheaper Drugs
The main patent on semaglutide, the active ingredient in blockbuster drugs like Ozempic and Wegovy developed by Novo Nordisk, expires in India on 20 March.
As soon as the protection lapses, Indian pharmaceutical companies are expected to launch their own lower-priced versions. One of the most significant outcomes of the patent expiry is expected to be a reduction in treatment costs. Currently, semaglutide-based therapies remain relatively expensive, which limits their widespread adoption.
With multiple companies entering the market, competition is likely to push prices downward. Lower prices could make semaglutide therapy more accessible to a larger population of patients dealing with diabetes and obesity in India. The availability of affordable generics may also encourage doctors to prescribe the drug more frequently, particularly for patients who previously could not afford the branded versions.
Rising Demand for Diabetes and Obesity Treatments
The anticipated surge of semaglutide generics comes at a time when India is witnessing a sharp rise in metabolic disorders. The country has one of the largest populations of people living with diabetes, and rates are further increasing rapidly.
This growing disease burden has created strong demand for effective and accessible therapies that can manage blood sugar levels while also helping patients control body weight.
GLP-1 receptor agonists such as semaglutide have emerged as an important tool in tackling both conditions simultaneously. As awareness about these treatments grows among healthcare professionals and patients, demand is expected to increase further.
A Major Opportunity for the Indian Pharma Industry
For Indian pharmaceutical companies, the semaglutide patent expiry represents a significant commercial opportunity. The expanding market for diabetes drugs offers strong growth potential for manufacturers capable of producing high-quality generics. At the same time, intense competition among dozens of brands may lead to aggressive pricing strategies and marketing campaigns. Companies will likely focus on differentiation through branding, physician engagement, and improved delivery technologies.
A Turning Point for Patient Access
The mid-March 2026 patent expiry of semaglutide is widely seen as a pivotal moment for India's metabolic disease treatment market. The entry of more than 50 branded generics could dramatically expand treatment availability while bringing down costs for patients.
As competition increases and prices fall, millions of people living with diabetes may soon gain improved access to one of the most effective therapies currently available. For both patients and the pharmaceutical industry, the coming months could mark the beginning of a new phase in metabolic disease management in India. India's obesity treatment market is about to change dramatically. With the key patent on semaglutide expiring this week, cheaper generic versions of the blockbuster diabetes drug are set to flood the market, potentially transforming how millions of Indians manage obesity. Doctors, hospitals, telehealth firms and startups are already preparing for a surge in demand.
Disclaimer: This content, including advice, provides generic information only. It is in no way a substitute for a qualified medical opinion. Always consult a specialist or your own doctor for more information. NDTV does not claim responsibility for this information.
Track Latest News Live on NDTV.com and get news updates from India and around the world



