Coronavirus Vaccine Approval
-
{
- All
- News
- Videos
- Web Stories
-

'Bivalent' Booster For Omicron, Original Covid Strain Approved By UK
- Saturday September 3, 2022
- World News | Press Trust of India
The UK health authorities on Saturday approved a second "bivalent" vaccine to be used as a booster vaccine to target both the Omicron and original strain of COVID-19.
-
 www.ndtv.com
www.ndtv.com
-

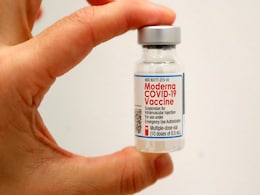
UK Approves An Updated Moderna Vaccine Which Targets Omicron Variant
- Monday August 15, 2022
- World News | Agence France-Presse
The UK's drug regulator said Monday it has approved an updated Moderna vaccine against the coronavirus that targets the Omicron variant as well as the original form.
-
 www.ndtv.com
www.ndtv.com
-

US Approves Covid Vaccine For Children 6 Months And Above
- Friday June 17, 2022
- World News | Agence France-Presse
The US Food and Drug Administration granted emergency authorization Friday for the use of Pfizer and Moderna Covid-19 vaccines in the youngest children, the final age group awaiting immunization in most countries.
-
 www.ndtv.com
www.ndtv.com
-

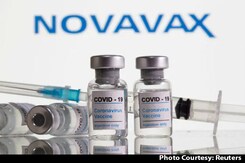
Immunisation Panel Approves Covovax Vaccine For 12-17 Age Group: Report
- Friday April 29, 2022
- India News | Asian News International
National Technical Advisory Group on Immunisation (NTAGI) on Friday approved Serum Institute of India's Covovax COVID-19 vaccine for the age group 12-17, said sources.
-
 www.ndtv.com
www.ndtv.com
-

Moderna Seeks US Approval For Covid Vaccine For Children Under 6
- Thursday April 28, 2022
- World News | Agence France-Presse
US biotech firm Moderna said Thursday it had submitted a request for an emergency use authorization in the United States for its Covid vaccine for children aged six months to under six years.
-
 www.ndtv.com
www.ndtv.com
-

Drug Firm Hetero Gets Approval To Produce, Sell Sputnik Light Vaccine
- Monday March 21, 2022
- India News | Press Trust of India
Drug firm Hetero on Monday said its biologics arm has received approval from the Central Drugs Standard Control Organisation (CDSCO) to manufacture and sell Sputnik Light for restricted emergency use in India.
-
 www.ndtv.com
www.ndtv.com
-

Moderna Seeks Emergency Approval For Second Covid Booster For US Adults
- Friday March 18, 2022
- World News | Agence France-Presse
Moderna announced Thursday it had asked the United States drug regulator for emergency authorization for a second booster shot of the company's Covid-19 vaccine for all adults.
-
 www.ndtv.com
www.ndtv.com
-

US Is Considering Second Covid Booster For 65+
- Wednesday March 16, 2022
- World News | Agence France-Presse
Pfizer and BioNTech announced Tuesday they had formally asked the United States drug regulator for emergency approval of a second booster shot of their Covid vaccine for people aged 65 and older.
-
 www.ndtv.com
www.ndtv.com
-

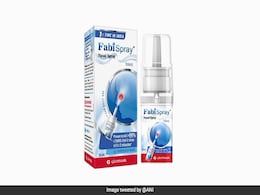
5-Point Guide To The New Nasal Spray For Covid Cleared By India
- Wednesday February 9, 2022
- India News | Reported by Vishnu Som
A nasal spray to treat adult COVID-19 patients has been launched in India after Mumbai-based developers Glenmark Pharmaceuticals received marketing clearances from the Drugs Controller General of India as part of an accelerated approval process.
-
 www.ndtv.com
www.ndtv.com
-

Pfizer To Seek US Approval For Covid Vaccine For Children Under 5: Report
- Wednesday February 2, 2022
- World News | Agence France-Presse
Pfizer and BioNTech will soon ask US regulators for emergency authorization for a Covid-19 vaccine for children aged five and under, US media reported Tuesday.
-
 www.ndtv.com
www.ndtv.com
-

'Bivalent' Booster For Omicron, Original Covid Strain Approved By UK
- Saturday September 3, 2022
- World News | Press Trust of India
The UK health authorities on Saturday approved a second "bivalent" vaccine to be used as a booster vaccine to target both the Omicron and original strain of COVID-19.
-
 www.ndtv.com
www.ndtv.com
-

UK Approves An Updated Moderna Vaccine Which Targets Omicron Variant
- Monday August 15, 2022
- World News | Agence France-Presse
The UK's drug regulator said Monday it has approved an updated Moderna vaccine against the coronavirus that targets the Omicron variant as well as the original form.
-
 www.ndtv.com
www.ndtv.com
-

US Approves Covid Vaccine For Children 6 Months And Above
- Friday June 17, 2022
- World News | Agence France-Presse
The US Food and Drug Administration granted emergency authorization Friday for the use of Pfizer and Moderna Covid-19 vaccines in the youngest children, the final age group awaiting immunization in most countries.
-
 www.ndtv.com
www.ndtv.com
-

Immunisation Panel Approves Covovax Vaccine For 12-17 Age Group: Report
- Friday April 29, 2022
- India News | Asian News International
National Technical Advisory Group on Immunisation (NTAGI) on Friday approved Serum Institute of India's Covovax COVID-19 vaccine for the age group 12-17, said sources.
-
 www.ndtv.com
www.ndtv.com
-

Moderna Seeks US Approval For Covid Vaccine For Children Under 6
- Thursday April 28, 2022
- World News | Agence France-Presse
US biotech firm Moderna said Thursday it had submitted a request for an emergency use authorization in the United States for its Covid vaccine for children aged six months to under six years.
-
 www.ndtv.com
www.ndtv.com
-

Drug Firm Hetero Gets Approval To Produce, Sell Sputnik Light Vaccine
- Monday March 21, 2022
- India News | Press Trust of India
Drug firm Hetero on Monday said its biologics arm has received approval from the Central Drugs Standard Control Organisation (CDSCO) to manufacture and sell Sputnik Light for restricted emergency use in India.
-
 www.ndtv.com
www.ndtv.com
-

Moderna Seeks Emergency Approval For Second Covid Booster For US Adults
- Friday March 18, 2022
- World News | Agence France-Presse
Moderna announced Thursday it had asked the United States drug regulator for emergency authorization for a second booster shot of the company's Covid-19 vaccine for all adults.
-
 www.ndtv.com
www.ndtv.com
-

US Is Considering Second Covid Booster For 65+
- Wednesday March 16, 2022
- World News | Agence France-Presse
Pfizer and BioNTech announced Tuesday they had formally asked the United States drug regulator for emergency approval of a second booster shot of their Covid vaccine for people aged 65 and older.
-
 www.ndtv.com
www.ndtv.com
-

5-Point Guide To The New Nasal Spray For Covid Cleared By India
- Wednesday February 9, 2022
- India News | Reported by Vishnu Som
A nasal spray to treat adult COVID-19 patients has been launched in India after Mumbai-based developers Glenmark Pharmaceuticals received marketing clearances from the Drugs Controller General of India as part of an accelerated approval process.
-
 www.ndtv.com
www.ndtv.com
-

Pfizer To Seek US Approval For Covid Vaccine For Children Under 5: Report
- Wednesday February 2, 2022
- World News | Agence France-Presse
Pfizer and BioNTech will soon ask US regulators for emergency authorization for a Covid-19 vaccine for children aged five and under, US media reported Tuesday.
-
 www.ndtv.com
www.ndtv.com